Private Mounjaro prescriptions, available now in Chislehurst. No referral. No waiting list.


Most people who start Mounjaro say the same thing a few weeks in: they wish they’d looked into it sooner.
Not because the medication is right for everyone — it isn’t. But because the process of finding out whether it’s right for you turned out to be far simpler than they expected. A conversation. An eligibility check. A clinical consultation. And, where appropriate, a prescription that can be in your hands within days.

Mounjaro (tirzepatide) is a prescription weight loss injection that works on the biology of hunger directly. It’s available at both Chislehurst Pharmacy Group locations without a GP referral and without a waiting list.
In Eli Lilly’s SURMOUNT-1 clinical trial, participants on the 15mg dose lost an average of 20.9% of their body weight over 72 weeks. Not through calorie restriction alone — through a medication that changes how the body regulates appetite and satiety at a hormonal level.
If you’ve been considering it and haven’t yet taken the next step, this article covers what you need to know.
Sustained weight loss is genuinely difficult, and the reason has less to do with discipline than most people assume.
When calorie intake drops, the body responds physiologically: hunger hormones increase, metabolism adjusts downward, and the signals that communicate fullness after eating become less effective. The longer a calorie deficit is maintained, the stronger these compensatory responses tend to become. For many people, this is the point at which progress stalls — not because they’ve stopped trying, but because the body is actively working against the deficit.

Mounjaro is a dual GIP and GLP-1 receptor agonist. It mimics two hormones the body naturally produces to regulate appetite, blood sugar, and the sensation of fullness after eating. Rather than suppressing hunger through stimulants, it works at the hormonal level — changing the signals the brain receives about food.
In practice, most patients describe a quietening of the constant background pull towards food. Portions feel satisfying at smaller sizes. The impulse to snack between meals fades. The effect is gradual rather than immediate, and it varies between individuals but for the majority of patients in clinical trials, it was meaningful.

The SURMOUNT trial programme represents the most extensive research conducted on a weight loss medication to date. The key findings are worth understanding clearly.
SURMOUNT-1 followed adults with obesity, or who were overweight without type 2 diabetes, over 72 weeks. On the 15mg dose, participants lost an average of 20.9% of total body weight.
For someone starting at 95kg, that is approximately 19–20kg. 91% of participants lost at least 5% of their body weight. 57% lost at least 20%.
SURMOUNT-3 examined patients who had already achieved weight loss through an intensive lifestyle programme and then continued with either Mounjaro or a placebo.
The Mounjaro group lost a further 21.1% beyond their initial reduction — a combined average of 26.6% from study entry.
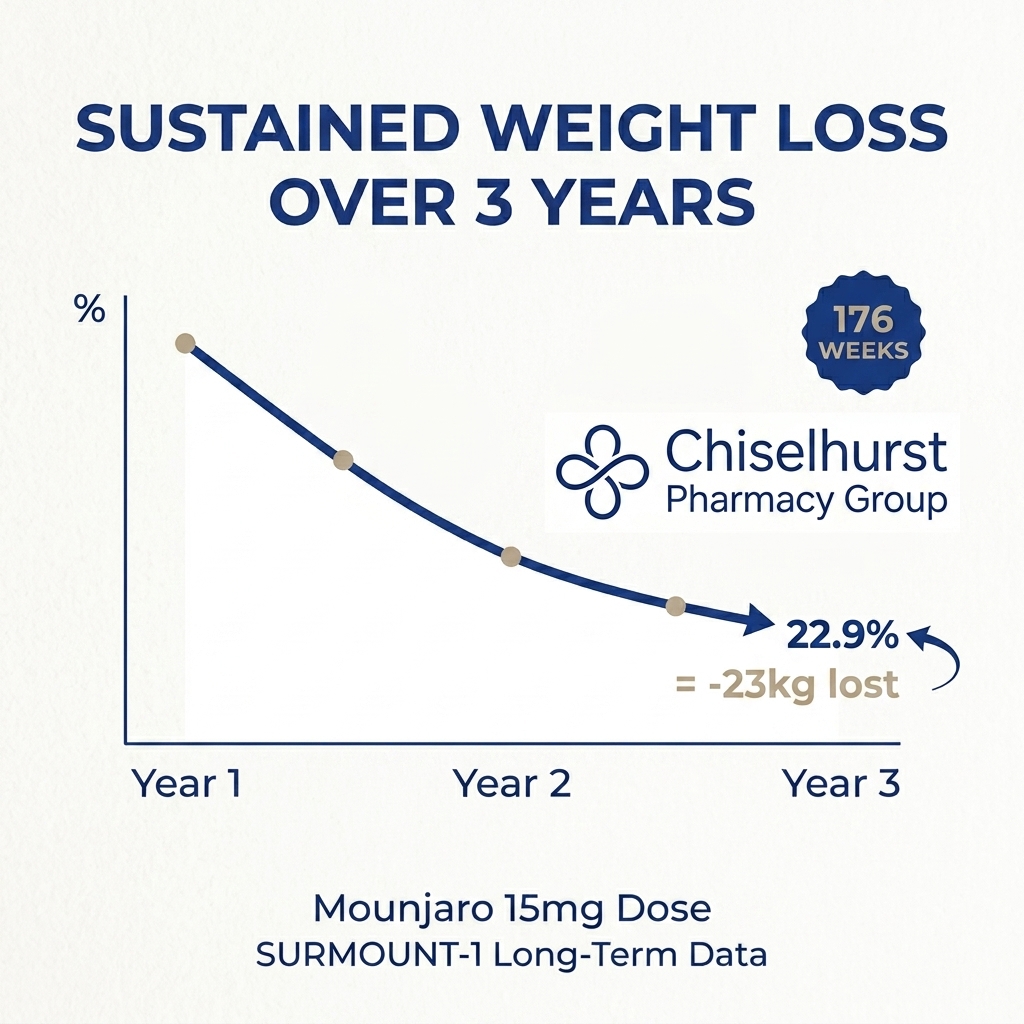
In late 2024, Eli Lilly published three-year follow-up data from SURMOUNT-1, showing 22.9% sustained weight loss at 176 weeks on the 15mg dose.
SURMOUNT-5 was a direct head-to-head comparison with Wegovy (semaglutide). Mounjaro produced 47% greater relative weight loss on average — 20.2% compared to 13.7% over 72 weeks.
These are average outcomes from controlled clinical trials. Individual results will vary depending on starting weight, the dose reached, adherence, diet, and activity levels. The figures give a reasonable picture of what is achievable for many patients — not a guarantee for every patient.

At Chislehurst Pharmacy Group, you are likely to be eligible if:
If you qualify, you’ll move into a full clinical consultation to discuss whether Mounjaro is appropriate given your specific health history, circumstances, and goals. If it isn’t the right option for you, Sumeet will explain why and discuss what alternatives may be worth considering.

Weeks 1–4: Starting dose Treatment begins at 2.5mg, administered as a once-weekly subcutaneous injection. The low starting dose allows the body to adapt gradually. Some patients notice mild nausea or a reduced appetite early on. Many notice very little change at this stage. Both responses are normal — the medication is working at a hormonal level, and visible results follow over time rather than immediately.
Weeks 5–8: Dose escalation The dose steps up to 5mg, and potentially 7.5mg, depending on tolerance. For most patients this is the stage at which appetite suppression becomes noticeably more pronounced — meals feel satisfying at smaller portions, and the urge to snack diminishes. Weight loss typically begins to accelerate during this period.
Weeks 9–12: Finding the right dose Sumeet will continue adjusting your dose based on how you’re responding and tolerating the medication. Some patients do well at 10mg. Others progress to 15mg. The aim is always the highest dose you tolerate comfortably, as this is associated with the best outcomes in trial data.
Most patients describe the first month as a period of adjustment, with the second and third months bringing more visible results — on the scales and in how clothes fit and how they feel day to day.
The most commonly reported side effects are gastrointestinal: nausea, loose stools, constipation, and reduced appetite. In Eli Lilly’s trial data, these were predominantly mild to moderate and concentrated during dose escalation. They tend to ease as the body adjusts to each dose level.
Steps that help manage symptoms:
More serious side effects are uncommon. Mounjaro carries a warning regarding thyroid tumours observed in animal studies — this is why it is not prescribed to anyone with a personal or family history of medullary thyroid carcinoma or MEN 2 syndrome. Pancreatitis has been reported rarely in patients using GLP-1 receptor agonists.
If you experience severe or persistent abdominal pain, or prolonged nausea and vomiting that doesn’t settle, contact Sumeet’s team promptly or seek medical attention.
Every patient goes through a full medical history review before any prescription is issued. Mounjaro is only prescribed where it is clinically appropriate to do so.
Chislehurst Pharmacy Group operates from two branches, both offering the same clinical service and the same standard of care from Sumeet Banker.
Pond Pharmacy 59 High St, Chislehurst, BR7 5AF 020 8467 3158 Mon–Fri: 9am–6pm · Sat: 9am–1pm
Chislehurst Pharmacy 59 Chislehurst Rd, Chislehurst, BR7 5NP 020 8295 0017 Mon–Fri: 9am–6pm · Sat: 9am–1pm
No GP referral is required at either location. Same-day and next-day appointments are usually available. Patients from across the London Borough of Bromley — including Bromley, Orpington, Sidcup, and Bexley — are welcome at whichever branch is more convenient.
The clinical service includes initial eligibility assessment, full consultation with Sumeet, ongoing dose reviews, and prescription management throughout your treatment.
If you’ve been thinking about Mounjaro and haven’t yet had a conversation about it, the process of finding out whether it’s suitable for you is straightforward.
Call either branch or book online. Sumeet’s team will run through your eligibility, take a full medical history, and give you a clear answer. If Mounjaro is appropriate for you, you’ll leave with a treatment plan and a prescription. If it isn’t, you’ll know exactly why — and what the options are.
Most patients say the hardest part was deciding to make the call.
Find out if Mounjaro is right for you.
Book your eligibility consultation at either Chislehurst Pharmacy Group location. Same-day and next-day appointments usually available. No GP referral needed.

Ready to take the next step?
Book your consultation at either Chislehurst Pharmacy Group location. Same-day and next-day appointments usually available.
Yes. Chislehurst Pharmacy Group offers Mounjaro on private prescription at both Pond Pharmacy on the High Street and Chislehurst Pharmacy on Chislehurst Road — no GP referral required. If your BMI is 30 or above, or 27 or above alongside a weight-related condition such as high blood pressure or type 2 diabetes, you can book an eligibility assessment directly with our pharmacy team. Most patients find out whether they qualify within the first consultation, and where appropriate, treatment can begin immediatley.
In Eli Lilly’s SURMOUNT-1 clinical trial, adults on the 15mg dose lost an average of 20.9% of their total body weight over 72 weeks. For someone starting at 95kg, that is approximately 19–20kg. Ninety-one percent of participants lost at least 5% of their body weight, and 57% lost 20% or more. These are average outcomes from a controlled clinical trial — individual results will vary based on starting weight, the dose reached, diet, activity, and other factors. Three-year follow-up data published in 2024 showed that results were maintained over time for participants who continued treatment.
The most common side effects are nausea, loose stools, constipation, and reduced appetite — particularly during dose increases. In clinical trials these were predominantly mild to moderate and tended to settle as patients adjusted to each dose level. Eating smaller, more frequent meals and avoiding high-fat foods in the early weeks reduces symptoms for most people. Sumeet’s team monitors you throughout treatment and will adjust your dose if side effects become a concern.
The two medications work through different mechanisms. Wegovy targets one hormone pathway (GLP-1). Mounjaro targets two — GIP and GLP-1. In the SURMOUNT-5 head-to-head trial, Mounjaro produced 47% greater relative weight loss on average: 20.2% versus 13.7% over 72 weeks. These are trial averages, and individual responses vary. If you have previously tried Wegovy, your consultation is a good opportunity to discuss your experience and whether Mounjaro is a suitable next step.
Mounjaro is MHRA-approved and has been studied in large-scale clinical trials. It is not suitable for people with a personal or family history of medullary thyroid carcinoma or Multiple Endocrine Neoplasia syndrome type 2, and is not prescribed during pregnancy or breastfeeding. Before any prescription is issued our trained team conducts a thorough review of your medical history to identify any contraindications. Treatment is only offered where it is clinically appropriate for the individual patient.